APIS
| Active Pharmaceutical Ingredient | CAS NO. | Molecular Formula | Definition | Structural Formula |
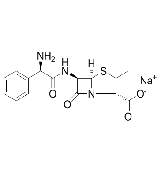
| Ampicillin Sodium | 69-52-3 | C16H18N3NaO4S | 4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, [6-(aminophenylacetyl)amino]-3,3-dimethyl-7-oxo-, monosodium salt, [2S-[2α,5α,6β(S*)]]-; Monosodium D-(−)-6-(2-amino-2-phenylacetamido)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate |
|
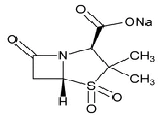
| Sulbactam Sodium | 69388-84-7 | C8H10NNaO5S | 4-Thia-1-azabicyclo[3.2.0]heptane-2-carboxylic acid, 3,3-dimethyl-7-oxo-, 4,4-dioxide, sodium salt, (2S-cis)-; Sodium (2S,5R)-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate 4,4-dioxide |
|
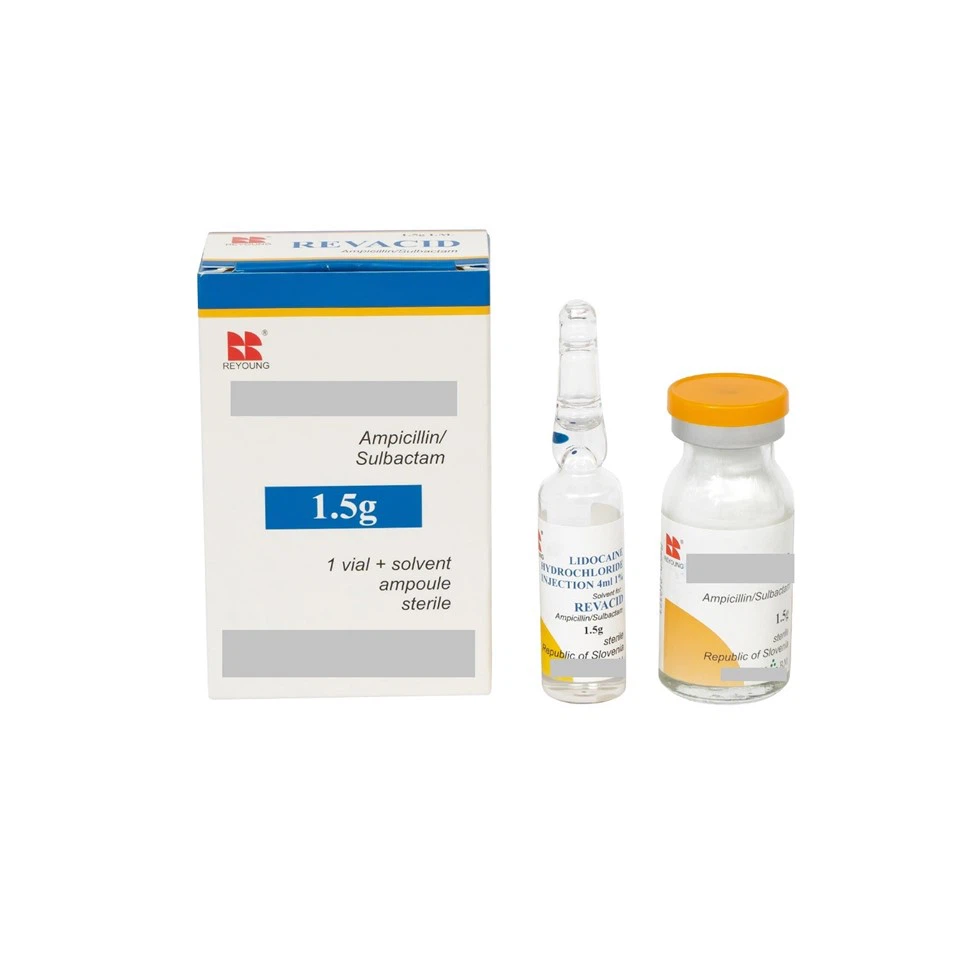
Products Specications
| Products | Specification | Vomume of vials | Packing form |
| Ampicillin Sodium and Sulbactam Sodium for Injection 0.75g,1.5g,3g | USP | 7ml mould vials | 10vials/box |
| 1vials/box | |||
| 10ml mould vials/ 10ml tube vials | 50vials/box | ||
| 10vials/box | |||
| 1vial/box | |||
| 1+1lidocaine/box | |||
| 12ml mould vials | 10vials/box | ||
| 1vial/box | |||
| 15ml mould vials | 25vials/box | ||
| 10vials/box | |||
| 1+1WFI/box | |||
| 20ml mould vials | 10vials/box | ||
| 1vial/box | |||
| 1+1WFI/box |
Composition:
Each vial of 750mg contains:
-Ampicillin Sodium equivalent to 500mg Ampicillin and Sulbactam Sodium equivalent to 250mg Sulbactam.
Each vial of 1.5g contains:
-Ampicillin Sodium equivalent to 1g Ampicillin and Sulbactam Sodium equivalent to 500mg Sulbactam.



Dosage And Administration
Ampicillin Sodium And Sulbactam Sodium For Injection is indicated for the treatment of infections due to susceptible strains of the designated microorganisms in the conditions listed below.
Skin and Skin Structure Infections caused by beta-lactamase producing strains of Staphylococcus aureus, Escherichia coli,* Klebsiella
spp.* (including K. pneumoniae*), Proteusmirabilis,* Bacteroides fragilis,* Enterobacter spp.,* and Acinetobacter calcoaceticus.*
Intra-Abdominal Infections caused by beta-lactamase producing strains of Escherichia coli,Klebsiella spp. (including K. pneumoniae*),
Bacteroides spp. (including B. fragilis), and Enterobacter spp.*
Gynecological Infections caused by beta-lactamase producing strains of Escherichia coli,* and Bacteroides spp.* (including B. fragilis*).
* Efficacy for this organism in this organ system was studied in fewer than 10 infections.
While AMPICILLIN/SULBACTAM is indicated only for the conditions listed above, infections caused by ampicillin-susceptible organisms are also amenable to treatment with AMPICILLIN/SULBACTAM due to its ampicillin content. Therefore, mixed infections caused by ampicillin-susceptible organisms and beta-lactamase producing organisms susceptible to AMPICILLIN/SULBACTAM should not require the addition of another antibiotic.
Appropriate culture and susceptibility tests should be performed before treatment in order to isolate and identify the organisms causing
infection and to determine their susceptibility to AMPICILLIN/SULBACTAM.
Therapy may be instituted prior to obtaining the results from bacteriological and susceptibility studies, when there is reason to believe the
infection may involve any of the beta-lactamase producing organisms listed above in the indicated organ systems. Once the results are
known, therapy should be adjusted if appropriate.
To reduce the development of drug-resistant bacteria and maintain effectiveness of AMPICILLIN/SULBACTAM and other antibacterial
drugs, AMPICILLIN/SULBACTAM should be used only to treat or prevent infections that are proven or strongly suspected to be caused
by susceptible bacteria. When culture and susceptibility information are available, they should be considered in selecting or modifying
antibacterial therapy. In the absence of such data, local epidemiology and susceptibility patterns may contribute to the empiric selection of
therapy.
Directions For Use
- General Dissolution Procedures: AMPICILLIN/SULBACTAM sterile powder for intravenous and intramuscular use may be reconstituted with any of the compatible diluents described in this insert. Solutions should be allowed to stand after dissolution to allow any foaming to dissipate in order to permit visual inspection for complete solubilization.
- Preparation for Intravenous Use: AMPICILLIN/SULBACTAM sterile powder in piggyback units may be reconstituted directly to the desired concentrations using any of the following parenteral diluents.
- Reconstitution of AMPICILLIN/SULBACTAM, at the specified concentrations, with these diluents provide stable solutions for the time periods indicated in the following table: (After the indicated time periods, any unused portions of solutions should be discarded.)
- OVERDOSE: If you miss a dose, use it as soon as you can. If it is almost time for your next dose, use only that dose. Do not use double or extra doses.
- SHELF LIFE: Three years.
- Note: Ampicillin Sodium And Sulbactam Sodium For Injection should be stored in a dry place below 30℃, away from light.
- When concomitant therapy with aminoglycosides is indicated, AMPICILLIN/SULBACTAM and aminoglycosides should be reconstituted and administered separately, due to the in vitro inactivation of aminoglycosides by any of the aminopenicillins.
- KEEP OUT OF REACH OF CHILDREN.



Hot Tags: ampicillin sodium and sulbactam sodium for injection, China ampicillin sodium and sulbactam sodium for injection manufacturers, suppliers, factory, vitamins solid preparations, powder for injection, furosemidum Injection, Meropenem For Injection, Metamizole Sodium Tablets, solid preparation